Only the U-235 isotope of uranium is fissionable. In natural uranium, U-235 is found in concentrations around 0.7%. The enrichment process increases the concentration of U-235 isotopes to a level of 3% to 5%. It is then usable as fuel for commercial nuclear power reactors. Currently, uranium enrichment is done with a gas centrifuge. Below is a detailed description of the enrichment process.

1. Delivery
Cylinders containing natural uranium, in the form of uranium hexafluoride (UF6), arrive at an enrichment facility, where they are inspected and sampled.
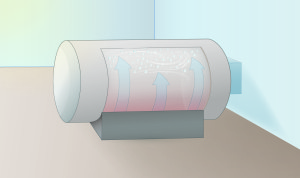
2. Transformed into a Gas
Each cylinder of UF6 is loaded into a huge autoclave where it is heated until the UF6 becomes a gas.
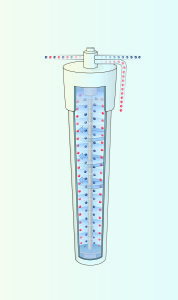
3. Gas Centrifuge
A gas centrifuge uses centrifugal force to concentrate the heavier U-238 molecules at the outer wall and lighter U-235 molecules toward the rotor center. The rotor, which contains the UF6 gas, spins at a high speed inside a vacuum casing. Gas circulation carries product (enriched with U-235 isotopes) and tails (depleted of U-235 isotopes) to opposite ends of the machine. Enrichment levels and capacity are increased by connecting centrifuges in series, called cascades. As the UF6 gas progresses through the cascade, the concentration of U-2325 is steadily increased until it reaches a level of up to 5%.
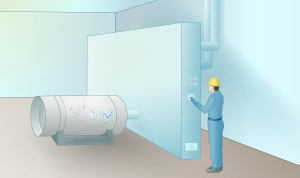
4. Transfer
At the product withdrawal facility, enriched UF6 is withdrawn from the cascade into 10-ton cylinders and sampled to ensure it meets specifications. It is later loaded into smaller 2.5-ton product of cylinders.
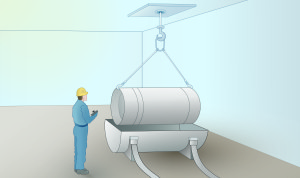
5. Ready for Shipment
The product cylinders are then loaded into sturdy protective containers for shipment to a nuclear fuel fabricator where the enriched uranium is converted into fuel assemblies for nuclear power reactors.
